 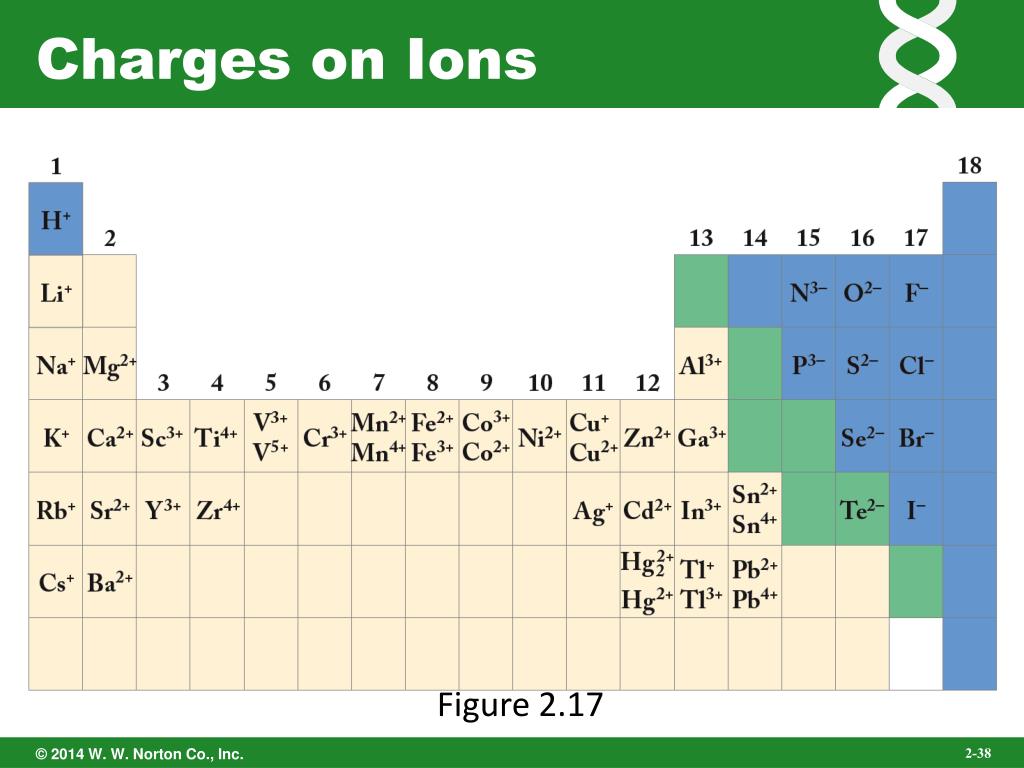
provide the ion name for the resultant ion.provide the ion symbol for the resultant ion, and.Consequently, the correct chemical formula for strontium fluoride is SrF 2.įigure 1. The alkali metals form ions with a +1 charge the alkaline earths, +2 and the halogens, –1.\) to determine the charge that will result upon its ionization, For one of them to form a compound with a halogen, twice as many halogen ions are needed to balance the +2 charge on the metal ions. All the remaining metal elements produce at least one ion with a charge of 2+. Ions formed from elements in Group IA of the periodic table have a 1+ charge. 'zero', In the middle of the row, we find silicon. From the right, the charge is minus one, minus two and minus three. From the left, the charge is plus one, plus two and plus three. This way we can figure out the ionic charge of a substance, just by finding it in the periodic table. The alkaline earths have ions with a charge of +2. The periodic table can help in predicting the type of ion that an element. This ion, the phosphide ion, gets a charge of minus three. These elements are metals that form oxides that have an earthy texture and yield alkaline solutions. By contrast, consider the alkaline earths in the second column on the left side of the table. Charges are balanced in a compound with the number of alkali metal ions equal to the number of halogen ions NaCl is such a compound. The alkali metals, on the other side of the periodic table, all readily lose one electron, so their ions possess a charge of +1. The halogens all tend to gain one electron, giving their ions a characteristic charge of –1. In the periodic table, elements in a column form analogous compounds because they have the same charge on their ions. The chemical behavior of the various elements is influenced more by the charge of their ion than by any other intrinsic property. Interactive periodic table with up-to-date element property data collected from authoritative sources. Table 1 shows three different charge states for copper: A positive ion has lost one or more electrons, whereas a negative ion has gained one or more electrons. Atoms with a charge imbalance are called ions. A neutral atom has precisely equal numbers of protons (+) and electrons (–). Of the three major subatomic particles, the negatively charged electron was first discovered by the English physicist Joseph Thomson in 1897.Īs the structure of atoms was probed, it was realized that these low‐mass particles occurred in a large “cloud” around the tiny nucleus, which contained almost all the mass of the atom. Quiz: Introduction to Oxidation-Reduction Reactions The periodic table (Figure 3.2 A Simple Periodic Table) can be used to determine which elements are metals and nonmetals.Introduction to Oxidation-Reduction Reactions.Well learn more about why this is the case in future chapters but for the time being if you can learn this trend. Common examples include sodium, potassium, calcium, chloride, and bicarbonate. Many normal substances exist in the body as ions. Ions with a negative charge are called anions. The mass spectrum of a sample shows the relative abundances of the ions on the y-axis and their m / z. Ions with a positive charge are called cations. A mass spectrometer ionizes atoms and molecules with a high-energy electron beam and then deflects the ions through a magnetic field based on their mass-to-charge ratios ( m / z. The elements at the end of the periodic table do not form ions. The latest release of the Periodic Table (dated ) includes the most recent abridged standard atomic weight values released by the IUPAC Commission on Isotopic Abundances and Atomic Weights (), compiled as part of the 2021 Table of Standard Atomic Weights 2021. An ion is an atom or group of atoms that has an electric charge. Quiz: Heat Capacities and Transformations Elements in the third to last column almost all form an anion with a 2- charge and elements living in the second to last column almost all form anions with a 1- charge.Quiz: Introduction to Organic Compounds. 
Quiz: Compounds with Additional Elements In an ionic compound, the cations and the anions combine in the smallest wholenumber ratio which mutually neutralizes the overall charge. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
